GE HealthCare's SIGNA MRI Portfolio Granted FDA Clearance
- Posted on February 20, 2026
- By Google News
- 3 Views
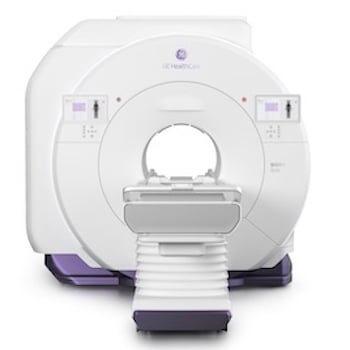
GE HealthCare's SIGNA MRI Portfolio Granted FDA Clearance

Feb. 19, 2026 — GE HealthCare recently announced 510(k) clearance of three new magnetic resonance (MR) innovations with the U.S. Food and Drug Administration (FDA): SIGNA Sprint with Freelium1, a 1.5T sealed magnet MRI system; SIGNA Bolt2, an advanced 3T MRI scanner; and SIGNA One3, an AI-driven ecosystem of workflow solutions designed to help reduce inefficiencies and support MRI exams from plan to scan and beyond.




