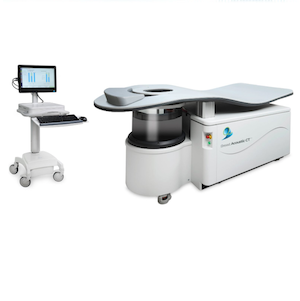
Breast Acoustic CT Scanner Receives FDA 510(k) Clearance
- Posted on March 13, 2026
- By Google News
- 4 Views
Breast Acoustic CT Scanner Receives FDA 510(k) Clearance

March 10, 2026 — QT Imaging Holdings has received U.S. Food and Drug Administration (FDA) 510(k) clearance for an updated configuration of its Breast Acoustic CT scanner, the company’s 3D ultrasound tomographic breast imaging system. The newly cleared enhanced configuration improves visualization and expands coverage of posterior breast tissue.




